Business Partnerships
Competitive Advantages
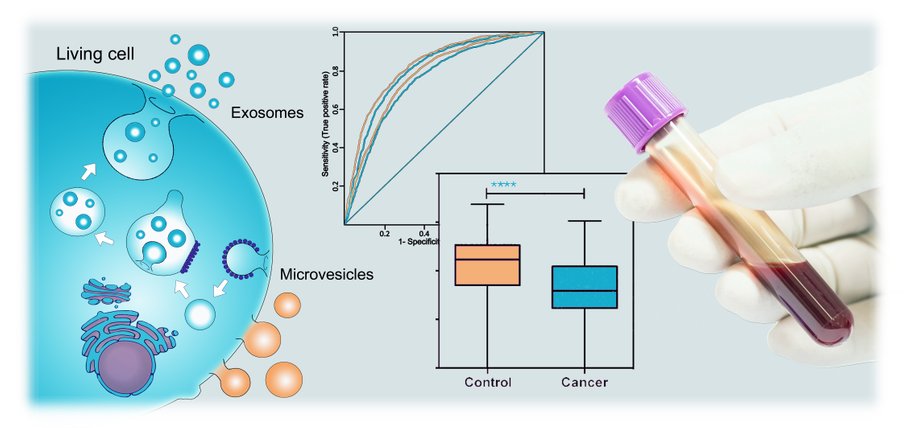
The research field of EVs has shown a great demand for commercial available technologies to analyse EVs for biomarkers. Currently, extensive and time consuming (> 24 hours) purification procedures is needed prior to analysis of single markers. NO OTHER existing technology phenotypes EVs in a multiplexed manner using unpurified material.
The EV Array consumes ONLY 10 – 100 µL SAMPLE, whereas purification procedures uses several milliliters. The EV Array is performed in multi-well cassettes in a HIGH-THROUGHPUT manner (up to 21 samples per slide), but is still easy to handle in the laboratory.
The use of microarray as a platform with spots of 1 nL volumes minimizes the cost of the EV Array as only small amounts of antibodies are needed.
The EV Array has been tested and optimized for various bodyfluids (plasma/serum, saliva, urine, ascites, sputum, bronchoalveolar lavage (BAL), ceresponial fluid, bone marrow, synovial fluid) together with cell culture supernatants.
Current Stage of Technology
The development of the EV Array started in 2011 and was published in Journal of Extracellular Vesicles (JEV) in 2013. In the developmental process all steps in the procedure were optimized including the microarray production and preanalytical quality control. Slides with various coatings has been tested together with buffer systems for printing, blocking and dilution as well as sample volumes, antibody concentrations, incubation time and temperature.
The current stage of the EV Array technology is a fully developed analysis optimized for a small-to-medium laboratory research scale.
To make the EV Array commercial available, a larger production of microarray slides including quality control is needed. Various commercial product combinations should be considered; predefined arrays or special customized arrays; kits (microarray slides, antibody solutions, buffers, cassettes); and additional services (fluorescence scanning and data treatment).
We are looking for:
- a partner for the ongoing development and optimization of the EV Array.
- a partner to make the EV Array commercial available in research and eventually clinical relations.
We will provide:
- Know-how and technical expertise in setting up the platform.
- We will be serving as a joint testing facility.
For further information regarding partnerships, please contact:
Valérie D Laurant
Commercialization Mananger
vkd@rn.dk
PARTNERSHIP